
Academics
OUR PROGRAMS
Earn a degree and make an impact in a healthcare-focused field.
A one-of-a-kind nursing degree-completion program
North Carolina will need 12,500 more nurses by 2032. To meet that talent gap, Northeastern’s Charlotte campus has added an innovative nursing degree-completion option to its Accelerated Bachelor of Science in Nursing (ASBN) program—the only one offered in North Carolina. This is a great path if you have an associate’s degree or at least 62 college credits, or if you have a bachelor’s degree in a non-nursing discipline.
All degree programs at Northeastern’s Charlotte campus
-
 Bachelor’s
Bachelor’sAccelerated Bachelor of Science in Nursing (ABSN)
Charlotte16 months -
 Master’s
Master’sApplied Behavior Analysis
Charlotte20 months -
 Professional Doctorate
Professional DoctorateEducation
Charlotte3-4 Years -
 Bachelor’s
Bachelor’sHealthcare Administration
Online5 years -
 Professional Doctorate
Professional DoctorateLaw and Policy
Charlotte3+ Years -
 Master’s
Master’sPublic Health
Charlotte1.5-5 Years -
 Master’s
Master’sSpeech Language Pathology
Charlotte2 Years -
 Master’s
Master’sSports Leadership
Charlotte12-18 months
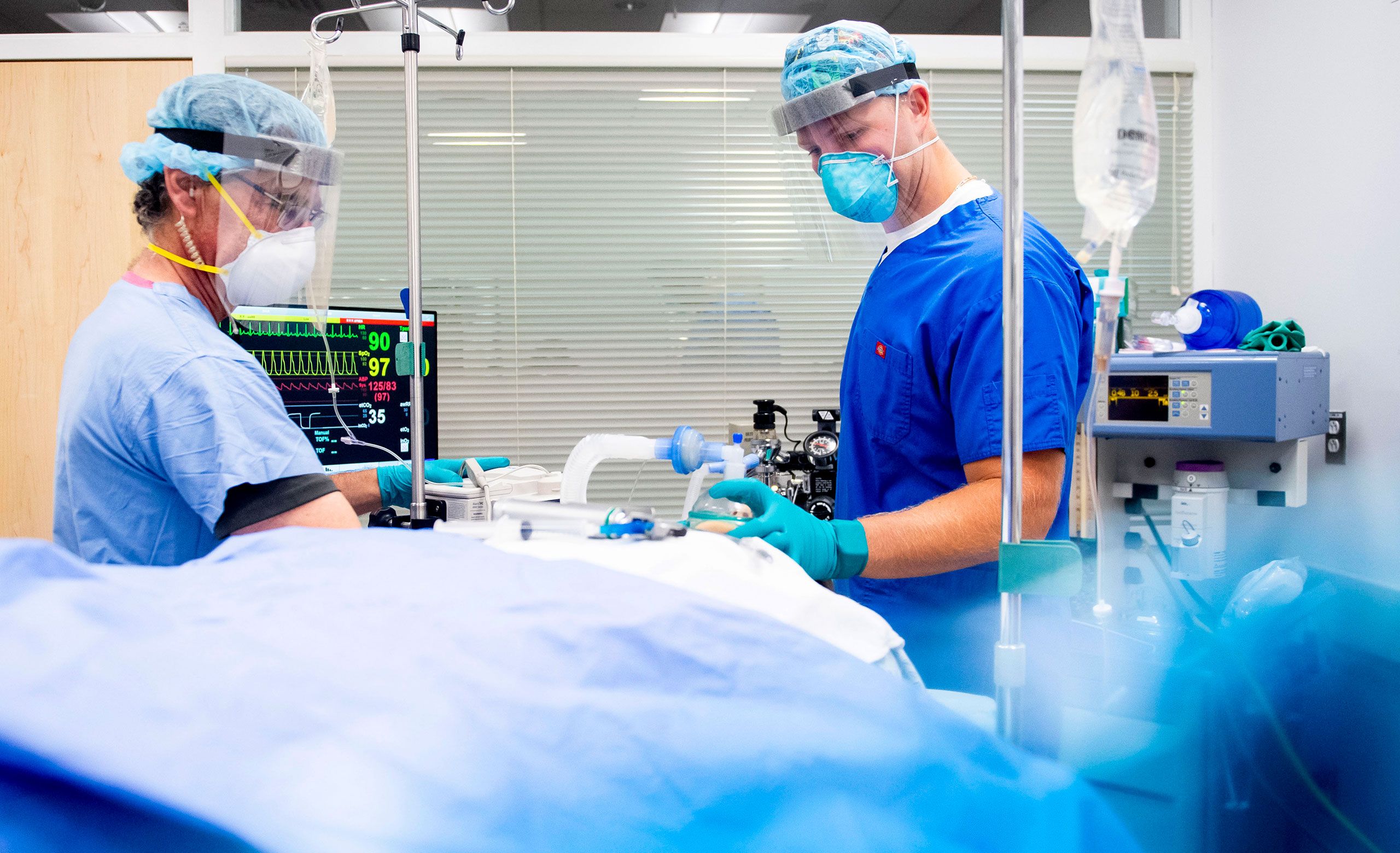
Community
A unique learning environment
The Center for Health Sciences anchors all healthcare work at Northeastern’s Charlotte campus: degree programs, research, and employer partnerships. As a student, you’ll be an integral part of this dynamic center, learning alongside Northeastern’s leading faculty experts as they collaborate with industry, government, and other academic institutions to solve pressing challenges in the healthcare sector.
TESTIMONIALS
Power of experience

“Students have access to an outstanding institution online yet with the personalized connection of a residency at the Charlotte campus. Plus, the Northeastern space is beautiful and smack in the middle of vibrant downtown Charlotte.”
Linda Kasarjian
Student in the doctorate in education program